
In comparison to other beta radiation-emitting nuclides, the electron is moderately energetic. The kinetic energy of the electron varies with an average of approximately 0.5 MeV and the remainder of the energy is carried by the nearly undetectable electron antineutrino. Phosphorus-32 is a beta emitter widely used in medicine and has a short half-life of 14.29 days and decays into sulfur-32 by beta decay as shown in this nuclear equation:ġ.709 MeV of energy is released during the decay. The diagram shows the type and energy of the emitted radiation, its relative abundance, and the daughter nuclides after decay. 137Cs is noted for a characteristic gamma peak at 661 KeV, but this is actually emitted by the daughter radionuclide 137mBa. The accompanying decay scheme diagram shows the beta decay of caesium-137. The 661 keV gamma peak associated with 137Cs is actually emitted by the daughter radionuclide. Both of these processes contribute to the copious quantities of beta rays and electron antineutrinos produced by fission-reactor fuel rods.Ĭaesium-137 decay scheme, showing it initially undergoes beta decay. Free neutrons also decay via this process. Β− decay commonly occurs among the neutron-rich fission byproducts produced in nuclear reactors. The virtual W − boson then decays into an electron and an antineutrino. At the quark level, W − emission turns a down quark into an up quark, turning a neutron (one up quark and two down quarks) into a proton (two up quarks and one down quark). The neutron turns into a proton through the emission of a virtual W − boson. This process is mediated by the weak interaction. 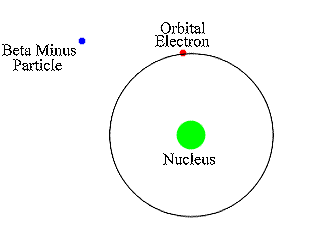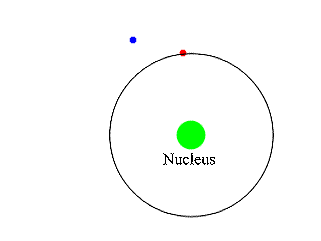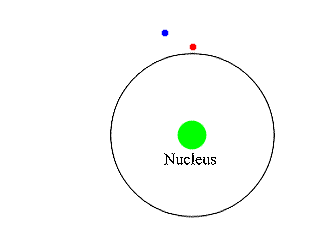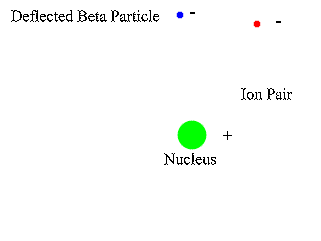 
Insert: in the decay of a free neutron, a proton, an electron (negative beta ray), and an electron antineutrino are produced.Īn unstable atomic nucleus with an excess of neutrons may undergo β − decay, where a neutron is converted into a proton, an electron, and an electron antineutrino (the antiparticle of the neutrino): An antineutrino (not shown) is always emitted along with an electron. A beta particle (in this case a negative electron) is shown being emitted by a nucleus. The higher the ionising effect, the greater the damage to living tissue, but also the lower the penetrating power of the radiation.īeta decay. īeta particles with an energy of 0.5 MeV have a range of about one metre in air the distance is dependent on the particle energy.īeta particles are a type of ionizing radiation and for radiation protection purposes are regarded as being more ionising than gamma rays, but less ionising than alpha particles. There are two forms of beta decay, β − decay and β + decay, which produce electrons and positrons respectively. Ī beta particle, also called beta ray or beta radiation (symbol β), is a high-energy, high-speed electron or positron emitted by the radioactive decay of an atomic nucleus during the process of beta decay. Beta radiation, consisting of electrons or positrons, is stopped by a thin aluminum plate, but gamma radiation requires shielding by dense material such as lead or concrete. Alpha radiation consists of helium nuclei and is readily stopped by a sheet of paper.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
